Since the nineteenth century, the world has seen remarkable inventions, innovations, and revolutionary advancements in energy and technology. A significant breakthrough occurred in 1917 with the discovery of ‘superconductivity’ by Dutch physicist Heike Kamerlingh Onnes. He observed that mercury exhibited unusual behavior at 4.2 Kelvin above absolute zero (-268.8°C), a phenomenon that captivated scientists and spurred extensive research. Despite its potential, the inherent difficulties in achieving superconductivity under practical conditions rendered the discovery largely unrealized for almost a century, despite some advancements.
This long-awaited discovery holds the promise of a new technological revolution. Imagine 100% efficient energy transmission, readily available maglev trains, and even smartphones that never need batteries. The critical question, however, has always been: “When will this become a reality?”
Recently, a team of physicists led by Ranga Dias at the University of Rochester achieved a significant breakthrough in this field. Their work could potentially provide the answer to “when” much sooner than anticipated, marking a truly revolutionary step forward. As Chris Pickard, a physicist from the University of Cambridge, enthusiastically states, “This is a landmark.”
Understanding Superconductivity: A Core Concept
To fully appreciate this potential revolution, let’s begin by understanding what superconductivity truly is.
Superconductivity describes a remarkable physical phenomenon where electric current flows through a material with absolutely no electrical resistance. In typical electrical circuits, materials possess resistivity, leading to energy loss, often as heat. However, in superconductors, materials exhibit zero resistance to electron flow, effectively eliminating energy dissipation in undesirable forms.
How does this occur? Under specific conditions of high pressure and extremely low temperatures, electrons overcome their natural repulsion and form pairs, known as Cooper pairs. In this unique, low-energy state, electrons exhibit quantum behavior, seemingly losing their individual identities. This allows them to move effortlessly through the atomic lattice without scattering, much like slipping through a crowd. Essentially, an electron slightly deforms the material’s structure, creating a pathway, and then pairs with another electron, collectively behaving as ‘superconductive electrons’ within ‘superconductors’. This mechanism is famously explained by the ‘BCS theory’.
Challenges in Achieving Practical Superconductivity
The primary obstacle preventing widespread practical application of superconductivity has been the extreme conditions it typically requires: pressures comparable to two-thirds of Earth’s core, combined with extremely low, non-ideal temperatures (far below 0°C). Materials only exhibit superconductive properties under these harsh environments. Consequently, ongoing research diligently explores various elements, compounds, and conditions in pursuit of achieving superconductivity at room temperature.
Decades after the initial discovery, significant progress was made in 1986 when physicists successfully lowered the required temperature to 30 Kelvin (K) using copper-oxide ceramics. This temperature is termed the material’s critical temperature (Tc). Numerous researchers and scientists subsequently investigated diverse materials and ceramics to find more suitable superconductors. By 1994, the critical temperature was pushed even higher, reaching 164 K in a mercury-based copper oxide compound under pressure.
Neil Ashcroft, a theoretical physicist at Cornell University, proposed an alternative material for achieving superconductivity above room temperature: metallic hydrogen. This substance forms when gaseous hydrogen, typically found in ambient atmospheres, solidifies under immense pressure—even exceeding that found at Earth’s core. Experiments involving hydrogen subjected to such extreme pressures within diamond anvil cells have been so intense they risked damaging the diamonds themselves.
However, in 2004, Ashcroft offered a refined approach, suggesting that pre-compressing hydrogen could enable it to become superconducting at higher temperatures and comparatively lower pressures. This innovative strategy proved successful.
A significant breakthrough occurred in 2015 when Mikhail Eremets and his team at the Max Planck Institute successfully addressed some challenges by utilizing sulfur hydride (H3S). They achieved superconductivity at 203 K and a pressure of 155 gigapascals (GPa). Further advancements, including the addition of lanthanum to hydrogen-rich compounds, raised the critical temperature to 250 K. Until recently, this represented the highest temperature achieved for superconductivity.
The Dawn of Room-Temperature Superconductors
In a groundbreaking development, Ranga Dias and his team recently discovered that introducing a third element, carbon, to hydrogen and sulfur compounds could significantly advance the quest for practical superconductors. Their initial experiments involved treating a sample at 148 GPa via electrical leads, where they observed superconductivity at 147 K. Remarkably, by increasing the pressure to 267 GPa, the material’s resistivity dropped to zero at an astonishing 287 K (approximately 15°C), a temperature easily achievable in winter conditions. This pivotal moment marked the achievement of room-temperature superconductivity in carbonaceous sulfur hydride, albeit under extreme pressures.
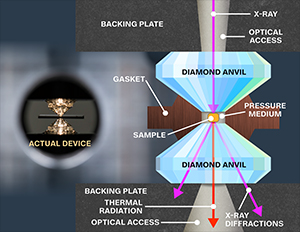
Dias and his collaborators are now focused on a crucial next step: finding ways to synthesize this superconducting material at significantly lower pressures. Dias draws an analogy, explaining, “Consider diamond: it’s a high-pressure form of carbon, yet today, we can grow it in laboratories using chemical deposition techniques. While it once demanded extreme pressure, we can now synthesize it; we might be able to achieve something similar with these new superconductors.”
Transformative Impact of Practical Superconductors
The realization of room-temperature superconductors at ambient pressures would trigger a profound ‘paradigm shift’ across the globe. As Ashkan Salamat, a co-author of the discovery from the University of Nevada Las Vegas, explains, “We currently thrive in a semiconductor-driven society. With this revolutionary technology, we could transition into a superconducting society where you’ll never need things like batteries again.”
Another remarkable property of superconductors is their ability to exclude magnetic fields, a phenomenon known as the Meissner effect. This characteristic enables the levitation of materials, laying the foundation for advanced transportation systems like levitating magnetic trains, commonly known as maglev trains.
Key potential applications of superconductivity include:
- Power grids capable of transmitting electricity with zero energy loss, eliminating the current dissipation of up to 200 million megawatt-hours (MWh) annually due to wire resistance.
- Innovative propulsion systems for levitated trains and other transportation methods, including advanced hyperloop concepts.
- Enhanced medical imaging and diagnostic tools, such as high-resolution MRI (Magnetic Resonance Imaging) and magnetocardiography.
- Significantly faster and more energy-efficient electronics for advanced digital logic circuits and memory devices.
And the potential applications continue to expand!
References:
- https://www.newscientist.com/article/2256953-first-room-temperature-superconductor-could-spark-energy-revolution/?utm_campaign=later-linkinbio-newscientist&utm_content=later-11009729&utm_medium=social&utm_source=instagram
- https://www.sciencemag.org/news/2020/10/after-decades-room-temperature-superconductivity-achieved
- https://www.rochester.edu/newscenter/rochester-sets-new-record-for-room-temperature-superconductor-455722/
Join our community by subscribing to our Weekly Newsletter to stay updated on the latest AI updates and technologies, including the tips and how-to guides. (Also, follow us on Instagram (@inner_detail) for more updates in your feed).
(For more such interesting informational, technology and innovation stuffs, keep reading The Inner Detail).





Pingback: Google claims to create Time Crystals inside a Quantum Computer: What does it mean to the world & us? – The Inner Detail